A curious thing about chemists is that we can find ourselves coalescing into groups based on which area of chemistry we find most interesting. While American physical chemist Gilbert N. Lewis was said to have declared, “Physical chemistry is everything that is interesting,” most organic chemists would certainly take issue with this perspective (and rightfully so) to say, “Organic chemistry is everything that is interesting!” The area of chemistry that most appeals to us, like beauty, is one of individual perspective and preference.

While each of us has our own “chemistry personality,” all of us chemists appreciate research accomplishments that take the field into a new realm through the discovery or creation of something that is truly awe-inspiring. We universally recognize research results for which, like gazing at a profoundly beautiful scene in the wilderness, our breath is taken away in reverence to the exposed mystery and complexity of nature that stands before us. Such paradigm shifts in discovery, often a result of years, even decades of hard work, are what make the mundane process of research rich with hope and possibility.
We are all very fortunate to be part of a field that has already taken human knowledge from the simple understanding of what makes up air and matter to understanding chemical bonding, identifying compounds that make up matter, and using that information to transform matter with precision and intention into food, medicines, pigments, plastics, and much more. Defining chemistry at a basic level, we might say that it is the study of matter and its transformations, but this simplified definition does not do justice to our “central science” and the myriad ways there are to approach the study of chemistry. Molecules range from very simple, like CO2, to extremely complex, such as vitamin B12 (Figure 1), and all are important. Research in chemistry spans efforts to understand the reactions of simple molecules, isolate complex molecules from nature, and synthesize even larger and more sophisticated structures.
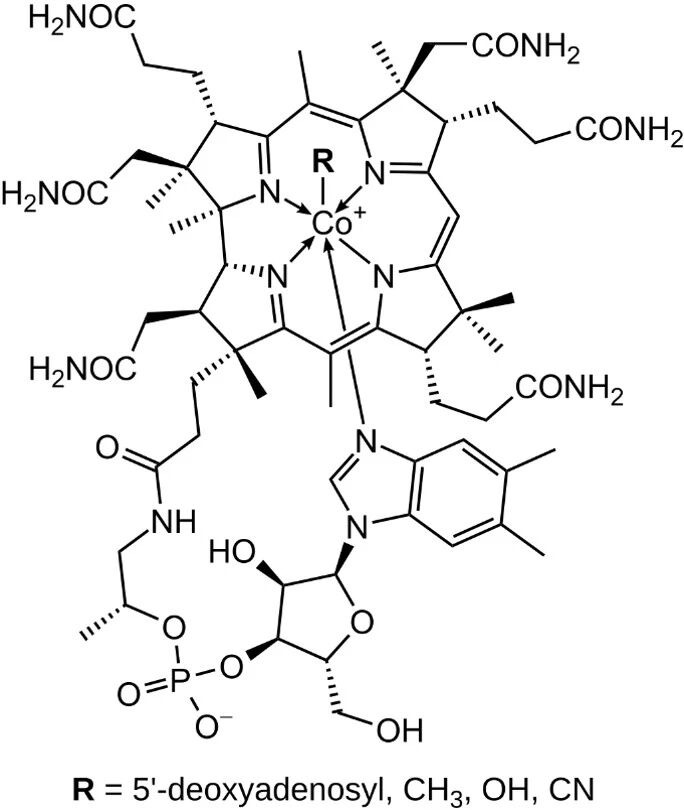
Organic synthesis holds a special place in the field of chemistry. This is a well-deserved honor, since organic synthesis allows chemists to replicate, modify, and create molecules that are used in nearly every area of modern life. The synthesis of molecules was described by Swiss chemist Albert Eschenmoser as, “The perhaps ‘most chemical’ activity of chemists…” American chemist, K. C. Nicolaou stated that, “synthesis is of paramount importance to our well-being, for through it we create new chemical entities (i.e. molecules) from which we derive our most precious material items.” He went on to say, “the flagship of organic synthesis is total synthesis,” and that this particularly challenging subfield of organic synthesis involving the building of natural products is both “a fine art and a precise science.” One of the first natural products to be synthesized in the laboratory was performed by German chemist, Friedrich Wöhler, almost two hundred years ago. This was the synthesis of urea and is often considered the birth of organic synthesis. Since then, thousands of more complex molecules have been synthesized, often requiring several dozen steps.
A molecule that stands out as a landmark in the development of total synthesis, and the field of chemistry in general, is vitamin B12. Determining the structure of vitamin B12 was itself a major accomplishment achieved by British chemist, Dorothy Hodgkin, who was awarded the Nobel Prize in 1964, “for her determinations by X-ray techniques of the structures of important biochemical substances,” which included penicillin and vitamin B12. And while the structure of vitamin B12 had been revealed, the total synthesis was so complex and extensive that it was considered the “Mount Everest” of synthetic chemistry at the time—a goal nearly impossible to obtain.
Its successful synthesis was ultimately completed in 1972 and became a key accomplishment in the history of synthetic chemistry. The synthesis required 12 years and close collaboration between Prof. Albert Eschenmoser’s research group at the Eidgenössische Technische Hochschule, Switzerland, and Prof. Robert B. Woodward’s laboratory at Harvard University, USA. It required around 100 total steps, done by trial and error. Prof. Eschenmoser later reflected, “the total synthesis of vitamin B12 provided a sort of proof that nowadays chemists can, in principle, synthesize any (low-molecular-weight) natural product structure, provided they are willing to invest the necessary time and effort.”

Under the joint leadership of Profs. Woodward and Eschenmoser, 91 post-doctoral researchers and doctoral students spanning 19 countries were intensely engaged in the effort to synthesize vitamin B12. Researchers and students worked extremely hard and felt great pride in being part of the complex and multifaceted effort. Dr. Srinivasa Ranganathan, a postdoctoral researcher working in Prof. Woodward’s laboratory noted, “On an average he [Prof. Woodward] used to put in 14–15 hours a day (Saturday half day); when I joined, he told me that he expected me to work for 100 hours a week! Towards the end it came to much more than that.”4 Another post-doctoral researcher in Prof. Woodward’s laboratory was CCS Chemistry Editorial Advisory Board member, Prof. Jean-Marie Lehn. Prof. Lehn visited Tsinghua University many years ago to give a talk on supramolecular chemistry, which was attended by Prof. Xi Zhang. In recounting the development of supramolecular chemistry, Prof. Lehn mentioned the total synthesis of vitamin B12 and pointed to the section of the molecule he worked on, which was indicated by a red circle, and he stated that, “this piece was synthesized by me, when I did post-doc research with R.B. Woodward at Harvard University.” Prof. Lehn’s pride in this achievement is especially notable, given that he received the Nobel Prize in 1987, along with Donald Cram and Charles Pedersen, for the synthesis of cryptands.
The total synthesis of “nature’s most beautiful cofactor,” vitamin B12, was a herculean effort to achieve, and it was only possible through a combination of creative approaches to the synthesis problem in combination with close, effective collaboration. Prof. Eschenmoser referred to the synthesis project between his and Prof. Woodward’s groups as a “collaborative competition,” and recounted, “The agreement was that neither of us should try to rush to the final goal to arrive there first. Both partners should fully exchange all the information that became available in the two laboratories along the way. This gives us the time and the opportunity on each side to explore the scientific problems of the project as extensively as possible. Within the decade that the collaboration lasted, there was indeed an enormous amount of information exchange going back and forth across the Atlantic.” In a fascinating and detailed retrospective of the process published in Tetrahedron in 2017, the authors extolled the uniqueness of the Eschenmoser–Woodward collaboration stating, “the mutual ingredient which both Woodward and Eschenmoser cherished for such a monumental collaborative project to succeed went well beyond the sheer strategy and numbers game. They required an absolute veracity, a mutual trust, to communicate and think as one but with the genius and brilliance of two.”
While a research feat on the scale of the total synthesis of vitamin B12 does not happen often, there are countless research groups and collaborations tackling complex chemical problems each day. We are proud to have some of that great work, representing the continued advancement of the beautiful field of chemistry, to report on in this month’s issue of CCS Chemistry. The issue starts with four Mini-Reviews: (1) supramolecular strategy for lithium batteries by Kai Liu et al., (2) fluorescence imaging for viral infections by Jong Seung Kim et al., (3) dynamic biomacromolecular modifications by Yan Huang, Jiarui Wu, Peng R. Chen, et al., and (4) photoresponsive polymer nanocomposites by Si Wu et al. In addition, there are three Communications and 18 Research Articles, covering copper-catalyzed oxidative coupling, C–H formylation of pyridines, organic electrosynthesis, construction of P-stereogenic centers, cation recognition, artificial water channel, covalent organic frameworks, lithium metal batteries, organic solar cells, white organic light-emitting diodes, upcycling of plastic wastes, CO2 conversion to C2+ products, and much more. A summary of each article in the issue is provided below.
Chemistry, and specifically the field of organic synthesis, continues to grow and evolve, perhaps faster than ever with the predictive and optimization capabilities of artificial intelligence (AI) and efficiencies introduced by autonomous robotic systems. Molecular engineering, first introduced in 1956 by German American chemist, Arthur R. von Hippel, as a way “…one builds materials from their atoms and molecules for the purpose at hand,” has become a field of its own, bringing together interdisciplinary teams to create new materials.7 While in the past, it may have taken 30 steps and a great deal of trial-and-error to synthesize a molecule with tailor-made structures and properties, with the help of AI, those 30 steps may be shortened to be ten or fewer steps. Such progress is already being realized, and there is much more to uncover with this compelling new approach to molecular synthesis.
With how fast technology is evolving and adding new ways to address complex problems, such as total synthesis, it is important to remind ourselves of the great tradition upon which chemistry has been built. Chemistry is and has always been a human endeavor, and the past (and present) is full of amazing researchers with colorful stories to share about their experiences, successes, and failures. Nearly all the great discoveries were also the result of a collaboration between intelligent, motivated individuals, such as Profs. Woodward and Eschenmoser, who were driven by a common curiosity. The quote by Issac Newton from 1675 is worth repeating and reminding ourselves of regularly, “if I have seen further, it is by standing on the shoulders of giants.” We owe these great figures in chemistry—both those in the past and those we have the fortune to interact with at present—our admiration and gratitude. Without them and their extraordinary discoveries in all fields of chemistry, we would not have such an expansive and exciting horizon in front of us to explore.
Prof. Dr. Xi Zhang
Editor-in-Chief
E-mail: xi@tsinghua.edu.cn
Dr. Donna J. Minton
Director of Publications, CCS
mail: donna.minton@chinesechemsoc.org